What is a Heat Pump?
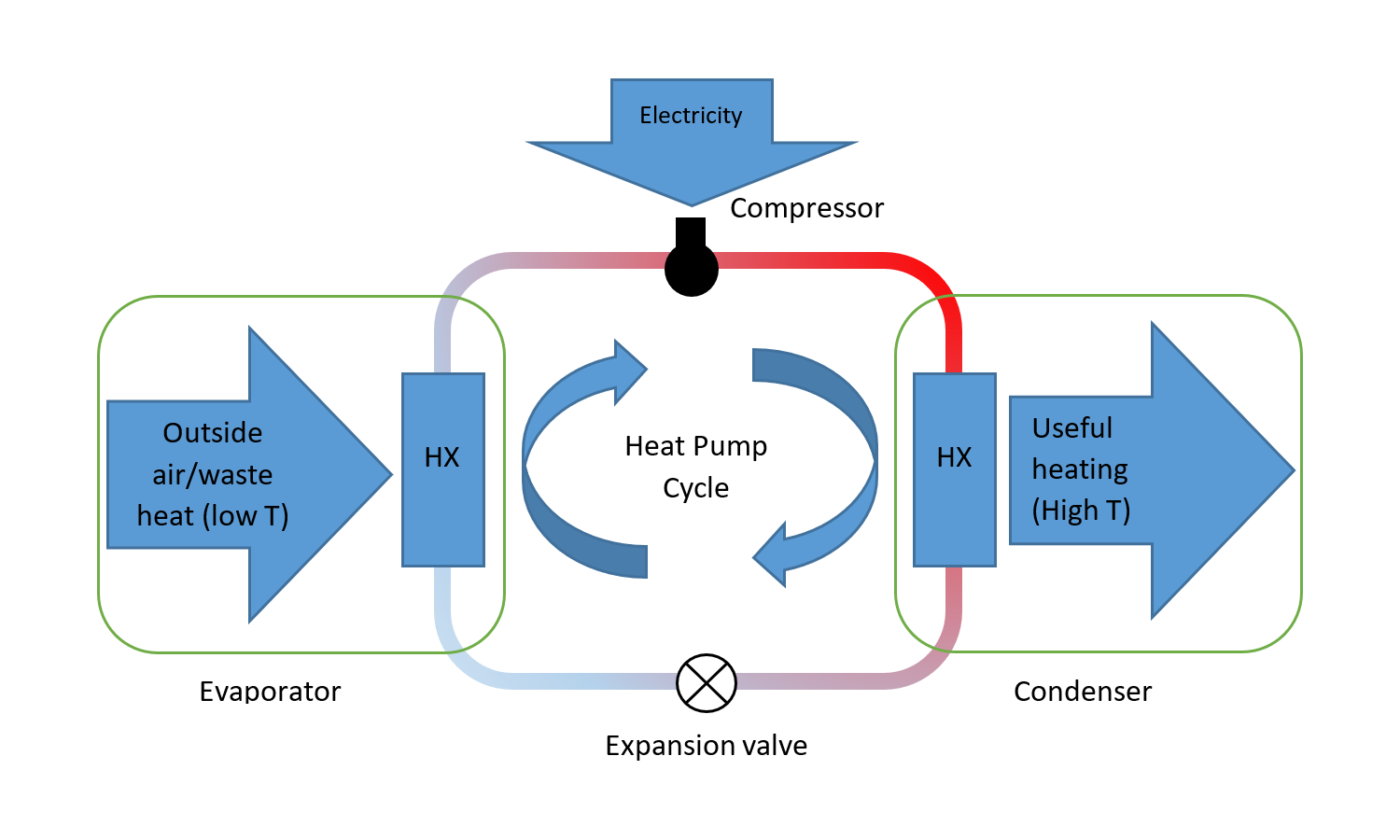
Figure 1. The heat pump cycle
Heat pumps have been around for as long as we have had refrigeration. If you have an air-conditioner that you use for heating in winter – also known as a reverse-cycle air-conditioner – then you are using a heat pump: The air-conditioner is extracting heat from the cool outside air and bringing it into the house via the heat pump cycle.
Key applications for heat pumps include:
- Air-conditioning: As noted above, an air-conditioner working reverse cycle mode is just a heat pump.
- Water heating: Heat pumps can be used for heating water for space heating or for domestic hot water heating. In Europe, the use of heat pumps to heat water for in-slab heating is very popular; some of these use the ground, rather than the air, as the heat source. Heat pump driven domestic hot water has been available in Australia for over 30 years.
- Industrial heat recovery: In industrial processes, heat pumps can be used to recycle lower temperature waste heat up to useful, higher temperature heat. Heat pumps can also be used in drying processes.
In this series of articles we’ll explore heat pump technology in more detail with a particular focus on carbon dioxide heat pumps, which are set to have a revolutionary impact on heat pump applications.
This article series is kindly contributed by Dr Paul Bannister, a thought leader and public speaker on energy and energy efficiency issues in Australia. For other articles by Dr Paul Bannister, please refer to our news section.
Nerd Space: What happens inside a heat pump?
Let’s start at the evaporator. The evaporator is typically a heat exchanger between the outside air and the refrigerant. The refrigerant enters the evaporator as a low pressure liquid. The boiling point of this liquid is cooler than the temperature of whatever we are extracting heat from – typically outside air. So as the outside air passes through the heat exchanger it makes the refrigerant evaporate. The energy required to evaporate the refrigerant is known as latent heat, and is all absorbed at one temperature, being the boiling point temperature – just like water boils at a fixed temperature of 100°C under normal conditions.
The refrigerant leaves the evaporator as a low pressure gas.
This low pressure gas enters the compressor where – as the name suggests – it is compressed. The compression process turns the cool, low pressure gas from the evaporator into a hot, high pressure gas. This gas enters the condenser.
The condenser is a heat exchanger between whatever it is we want to heat (which might be water or air) and the refrigerant. Because the refrigerant is under pressure, its boiling point temperature is higher than it was in the evaporator. Meantime, the water or air that we are heating is cooler than the refrigerant boiling point. This causes the refrigerant to condense and become liquid, releasing the latent energy to whatever we are heating.
The refrigerant leaves the condenser as a warm liquid. It then passes through the expansion valve where the pressure drops and as a result the refrigerant vaporises.
The magic of this process is that we transfer heat from a cold object to a hotter – something that can only happen because of the mechanical work input at the compressor.